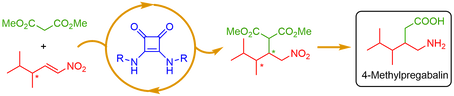Organocatalysis
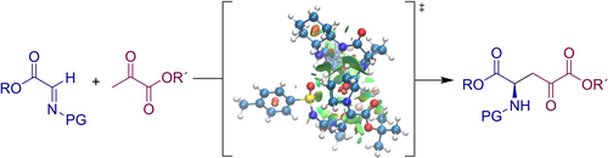
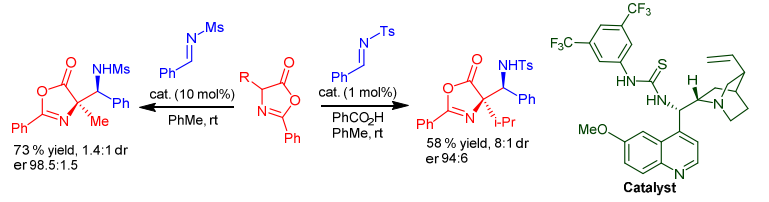
Enantioselective catalytic formation of polyfunctional molecules, such as non-natural amino acids, is important for the efficient production of many chiral compounds. To this end, we present here the synthesis and evaluation of the catalytic activity of bifunctional peptide–thiourea organocatalysts. These hybrid organocatalysts consist of Pro-Pro dipeptide and thiourea moiety connected via a 1,2-diaminocyclohexane unit. These catalysts promoted challenging Mannich reaction between α-imino esters and pyruvates, providing orthogonally protected oxo-glutamate derivatives. The N-tosyl-protected imino ester, as the most active imine, was required to compensate for the poor reactivity of pyruvates. DFT calculations, NMR, and CD spectroscopy help elucidate the mode of action of these catalysts. Configuration of the dipeptide Pro-Pro moiety is responsible for the sense of the stereoinduction of the Mannich reaction.
P. Čmelová, P. Šramel, B. Zahradníková, V. Modrocká, H. Szabados, M. Mečiarová, R. Šebesta, Eur. J. Org. Chem. 2022, e202200106. DOI: https://doi.org/10.1002/ejoc.202200106
P. Čmelová, P. Šramel, B. Zahradníková, V. Modrocká, H. Szabados, M. Mečiarová, R. Šebesta, Eur. J. Org. Chem. 2022, e202200106. DOI: https://doi.org/10.1002/ejoc.202200106
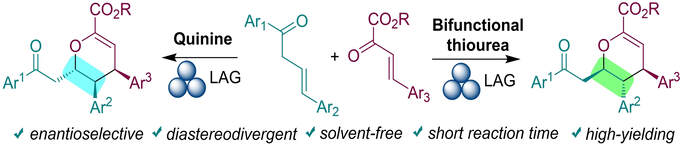
Chiral heterocycles occur in many compounds of interest, but their efficient synthesis is challenging. This study concerns the enantioselective and diastereoselective synthesis of densely substituted chiral pyran derivatives. Diastereodivergence of the oxa-Diels-Alder reaction is achieved by using either a bifunctional amino-thiourea or a monofunctional quinine organocatalyst under ball-milling conditions. Liquid-assisted grinding proves a highly efficient means of affording pyrans in high yield, with high enantiomeric purities and short reaction times.
T. Peňaška, V. Modrocká, K. Stankovianska, M. Mečiarová, E. Rakovský, R. Šebesta, ChemSusChem 2022, e202200028.
DOI: 10.1002/cssc.202200028.
T. Peňaška, V. Modrocká, K. Stankovianska, M. Mečiarová, E. Rakovský, R. Šebesta, ChemSusChem 2022, e202200028.
DOI: 10.1002/cssc.202200028.
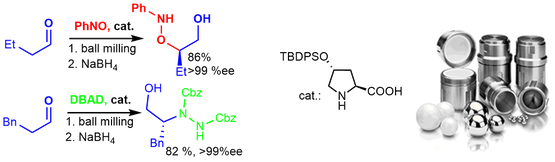
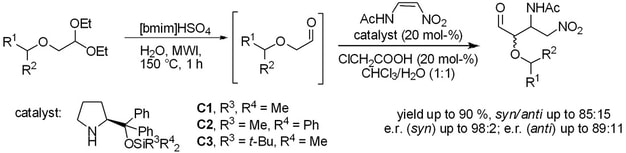
The synthesis of bifunctional N-sulfinylureas and thioureas with an appended pyrrolidine unit is presented. These organocatalysts were evaluated in Michael additions of aldehydes to nitroalkenes both under solvent-free conditions and in solution. The N-sulfinylurea catalyst was more efficient than the corresponding thiourea. For some substrates, enantioselectivities reached 98% ee. The stereogenic center on the sulfur did not have a considerable influence on the catalytic reactions. Under ball-milling conditions, the Michael adducts were obtained in good yields but with slightly lower enantiomeric purities than in solution. DFT calculations elucidated its mode of action and confirmed a dual activation mode, which combines enamine activation of aldehydes and hydrogen-bond activation of nitroalkenes.
V. Poláčková, D. Krištofíková, B. Némethová, R. Górová, M. Mečiarová, R. Šebesta, Beilstein J. Org. Chem. 2021, 17, 2629–2641.
DOI: https://doi.org/10.3762/bjoc.17.176
V. Poláčková, D. Krištofíková, B. Némethová, R. Górová, M. Mečiarová, R. Šebesta, Beilstein J. Org. Chem. 2021, 17, 2629–2641.
DOI: https://doi.org/10.3762/bjoc.17.176
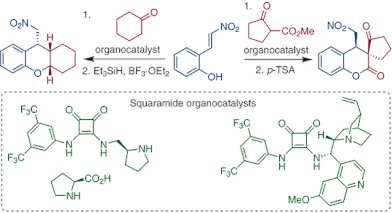
In this work, we describe the Michael addition–cyclization reaction of 2-(2-nitrovinyl)phenol with two different reactive Michael donors, which lead to chiral benzopyran derivatives. Specifically, bifunctional amino-squaramides with one or two chiral units in the side chains were evaluated as catalysts in these transformations. Furthermore, the utility of selected green solvents as reaction media for these processes was also tested. The best result was achieved with methyl-cyclopentanone-2-carboxylate as the Michael donor in ethyl (–)-l-lactate with quinine-based amino-squaramide as catalyst (yield 72%, dr >99:1, ee 99%).
E. Veverková, P. Molnosiová, R. Šebesta, SynOpen 2021, 5, 278-284.
DOI: 10.1055/s-0040-1719843
E. Veverková, P. Molnosiová, R. Šebesta, SynOpen 2021, 5, 278-284.
DOI: 10.1055/s-0040-1719843
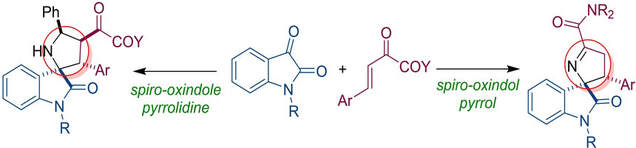
Spiro-oxindole pyrrolidines are formed by formal [3+2] cycloaddition between oxindole imines and unsaturated keto esters and keto amide. Keto amides also afford alternative products via enolate protonation and addition of nitrogen on the carbonyl group. Quinine-based squaramide organocatalyst was able to provide keto ester pyrrolidines in enantiomeric purity of up to e.r. 86 : 14.
T. Peňaška, V. Palchykov, E. Rakovský, G. Addová, R. Šebesta, Eur. J. Org. Chem. 2021, 1693-1703.
DOI: https://doi.org/10.1002/ejoc.202100022
T. Peňaška, V. Palchykov, E. Rakovský, G. Addová, R. Šebesta, Eur. J. Org. Chem. 2021, 1693-1703.
DOI: https://doi.org/10.1002/ejoc.202100022
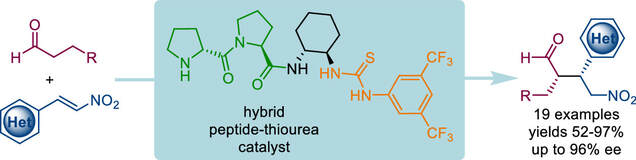
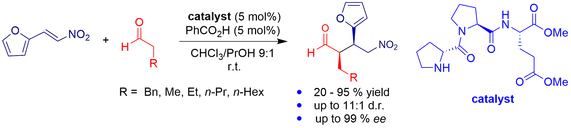
Bifunctional organocatalysis combining covalent and noncovalent activation is presented. The hybrid peptide–thiourea catalyst features a N-terminal proline moiety for aldehyde activation and a thiourea unit for electrophile activation. This catalyst effectively promotes asymmetric Michael additions of aldehydes to challenging but biologically relevant heterocycle-containing nitroalkenes. The catalyst can be used under solvent-free conditions. Spectroscopic and density functional theory studies elucidate the catalyst structure and mode of action.
P. Čmelová, D. Vargová, R. Šebesta, J. Org. Chem. 2021, 86, 581-592.
DOI: https://doi.org/10.1021/acs.joc.0c02251
P. Čmelová, D. Vargová, R. Šebesta, J. Org. Chem. 2021, 86, 581-592.
DOI: https://doi.org/10.1021/acs.joc.0c02251
Mechanochemical activation effectively mediated asymmetric organocatalytic domino Mannich addition followed by diastereoselective fluorination. The Mannich reactions of pyrazolones and to a lesser extent those of isoxazolones were effective under solvent-free ball-milling conditions. This reaction in combination with a chiral squaramide catalyst provided corresponding products in high yields and enantiomeric purities up to 99:1 e.r. and as a single diastereomer. DFT calculations revealed reasons for high diastereoselectivity.
D. Krištofíková, M. Mečiarová, E. Rakovský, R. Šebesta, ACS Sust. Chem. Eng. 2020, 8, 14417–14424.
DOI: https://doi.org/10.1021/acssuschemeng.0c04260
D. Krištofíková, M. Mečiarová, E. Rakovský, R. Šebesta, ACS Sust. Chem. Eng. 2020, 8, 14417–14424.
DOI: https://doi.org/10.1021/acssuschemeng.0c04260
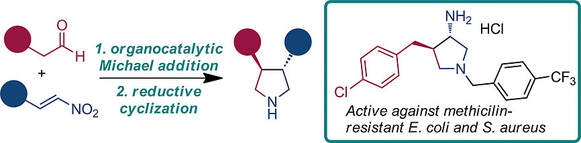
Chiral aliphatic heterocycles are important structural feature of many pharmaceutical agents. Antibiotic resistance is a serious medical problem, therefore new antibacterial compounds are urgently needed. Herein, we describe synthesis of a series of 3,4-disubstituted pyrrolidine derivatives via organocatalytic Michael addition followed by reductive cyclization. These compounds inhibited growth of standard as well as methicillin-resistant strains of Escherichia coli and Staphylococcus aureus.
L. Rodriguez, R. Fišera, B. Gaálová, K. Koči, H. Bujdáková, M. Mečiarová, R. Górová, H. Jurdáková, R. Šebesta, Eur. J. Org. Chem. 2020, 2565-2575.
DOI: https://doi.org/10.1002/ejoc.202000235
L. Rodriguez, R. Fišera, B. Gaálová, K. Koči, H. Bujdáková, M. Mečiarová, R. Górová, H. Jurdáková, R. Šebesta, Eur. J. Org. Chem. 2020, 2565-2575.
DOI: https://doi.org/10.1002/ejoc.202000235
Rapid improvements: Asymmetric organocatalysis can be efficiently combined with various green chemistry methodologies. Acceleration techniques, such as ball milling, flow, high pressure, or light, offer higher yields, faster reactions, easier product isolation, and new reactivities. More sustainable reaction media, such as ionic liquids, deep eutectic solvents, green solvent alternatives, and water, also improve the sustainability profile of organocatalytic reactions.
D. Krištofíková, V. Modrocká, M. Mečiarová, R. Šebesta, ChemSusChem 2020, 13, 2828-2858.
DOI: https://doi.org/10.1002/cssc.202000137
D. Krištofíková, V. Modrocká, M. Mečiarová, R. Šebesta, ChemSusChem 2020, 13, 2828-2858.
DOI: https://doi.org/10.1002/cssc.202000137
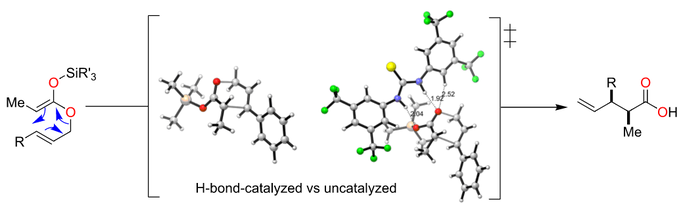
A range of chiral hydrogen-bond-donating organocatalysts was tested in the Ireland–Claisen rearrangement of silyl ketene acetals. None of these organocatalysts was able to impart any enantioselectivity on the rearrangements. Furthermore, these organocatalysts slowed down the Ireland–Claisen rearrangement in comparison to an uncatalyzed reaction. The catalyst-free reaction proceeded well in green solvents or without any solvent. DFT calculations showed that the activation barriers are higher for reactions involving hydrogen-donating organocatalysts and kinetic experiments suggest that the catalysts bind stronger to the starting silyl ketene acetals than to transition structures thus leading to inefficient rearrangement reactions.
D. Krištofíková, M. Mečiarová, E. Rakovský, R. Šebesta, ACS Sust. Chem. Eng. 2020, 8, 14417–14424.
DOI: https://doi.org/10.3762/bjoc.15.290
D. Krištofíková, M. Mečiarová, E. Rakovský, R. Šebesta, ACS Sust. Chem. Eng. 2020, 8, 14417–14424.
DOI: https://doi.org/10.3762/bjoc.15.290
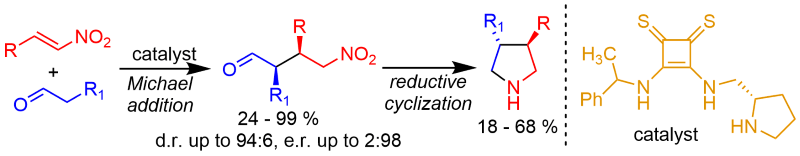
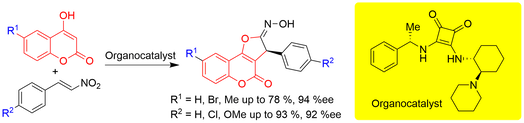
Bifunctional squaramide and thiosquaramide organocatalysts possessing pyrrolidine moiety for enamine activation catalyzed Michael additions of enolizable aldehydes to nitroalkenes and nitroacrylates. The Michael adducts were obtained in good yields, diastereomeric purities up to 94:6 and enantiomeric purities up to 2:98. Michael additions proceeded well also under solvent-free ball-milling conditions. These adducts were reductively cyclized to the corresponding chiral 3,4-disubstituted pyrrolidines.
K. Ormandyová, S. Bilka, M. Mečiarová, R. Šebesta, ChemistrySelect, 2019, 4, 8870-8875.
DOI: 10.1002/slct.201902652
K. Ormandyová, S. Bilka, M. Mečiarová, R. Šebesta, ChemistrySelect, 2019, 4, 8870-8875.
DOI: 10.1002/slct.201902652
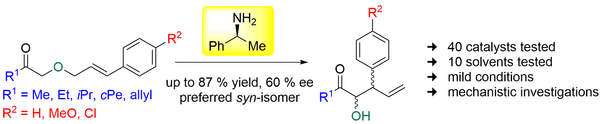
Organocatalytic [2,3]-Wittig rearrangement of allyloxyketones was investigated. Extensive screening of chiral amine organocatalysts showed that small pyrrolidine-based catalysts or simple phenylethylamine with the primary amino group were the most effective ones.
T. Peňaška; M. Meyer Mojzes; J. Filo; H. Jurdáková; M. Mečiarová; R. Šebesta, Eur. J. Org. Chem. 2019, 605-610; DOI: 10.1002/ejoc.201801697.
T. Peňaška; M. Meyer Mojzes; J. Filo; H. Jurdáková; M. Mečiarová; R. Šebesta, Eur. J. Org. Chem. 2019, 605-610; DOI: 10.1002/ejoc.201801697.
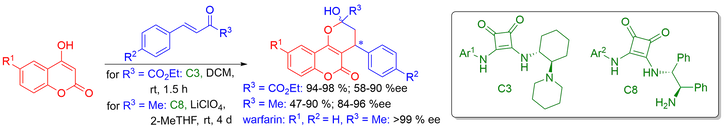
The catalytic efficiency of various amine-squaramides was tested in Michael/hemiketalization reactions of 4-hydroxycoumarines with two types of enones. The Michael addition of 4-hydroxycoumarin to 4-phenylbut-3-en-2-on directly produced chiral anticoagulant drug (S)-warfarin.
V. Modrocká, E. Veverková, M. Mečiarová, R. Šebesta, J. Org. Chem. 2018, 83, 13111-13120; DOI: 10.1021/acs.joc.8b01847.
V. Modrocká, E. Veverková, M. Mečiarová, R. Šebesta, J. Org. Chem. 2018, 83, 13111-13120; DOI: 10.1021/acs.joc.8b01847.
This work describes the squaramide-catalyzed Michael addition of 4-hydroxycoumarins to β-nitrostyrenes, followed by a cyclization reaction. In this way, a series of 2,3-dihydrofurocoumarin derivatives were synthetised in 47-93% yields and enantiomeric purities in the range of 78-94% ee.
V. Modrocká, E. Veverková, R. Baran, R. Šebesta, ChemistrySelect 2018, 3, 1466-1471; DOI: 10.1002/slct.201800147.
V. Modrocká, E. Veverková, R. Baran, R. Šebesta, ChemistrySelect 2018, 3, 1466-1471; DOI: 10.1002/slct.201800147.
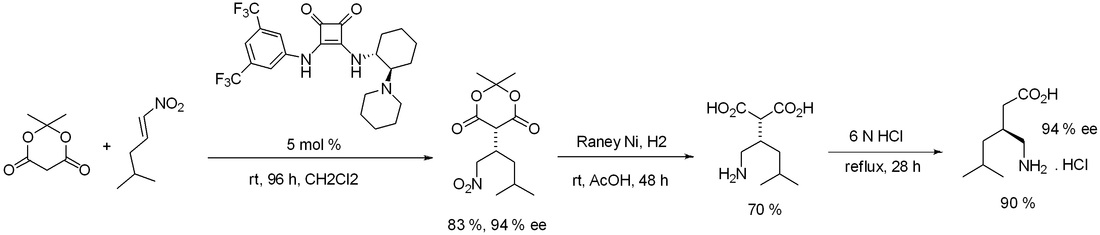
Asymmetric addition of dimethyl malonate to racemic nitroalkene, catalysed by a chiral squaramide catalyst, leads to an enantiomerically enriched adduct. The core structure is a key intermediate in the synthesis of 4-methylpregabalin.
D. Vargová, R. Baran, R. Šebesta, Beilstein J. Org. Chem. 2018, 14, 553-559; DOI:10.3762/bjoc.14.42.
D. Vargová, R. Baran, R. Šebesta, Beilstein J. Org. Chem. 2018, 14, 553-559; DOI:10.3762/bjoc.14.42.
Short-chain peptides have found many uses in asymmetric catalysis. In this work we investigate catalytic effect of new di- and tripeptide catalyst possessing a free secondary amine group, because they can activate carbonyl compounds via enamine formation and can successfully catalyze many reactions, Michael addition among them.
V. Poláčková, P. Čmelová, R. Górová, R. Šebesta, Monatsh. Chem. 2018, 149, 729-736; DOI: https://doi.org/10.1007/s00706-017-2126-2.
V. Poláčková, P. Čmelová, R. Górová, R. Šebesta, Monatsh. Chem. 2018, 149, 729-736; DOI: https://doi.org/10.1007/s00706-017-2126-2.
Application of aqueous media and solvent-free conditions have been assessed for asymmetric organocatalytic α-aminoxylation and hydrazination of aldehydes. Both sets of conditions considerably sped up these reactions, while keeping high enantioselectivity.
E. Veverková, V. Modrocká, R Šebesta, Eur. J. Org. Chem. 2017, 1191-1195; DOI: 10.1002/ejoc.201601357.
E. Veverková, V. Modrocká, R Šebesta, Eur. J. Org. Chem. 2017, 1191-1195; DOI: 10.1002/ejoc.201601357.
An efficient three-component organocatalytic cascade 1,3-dipolar cycloaddition reaction of in situ generated azomethine ylides with α,β-unsaturated esters is described. This reaction afforded a small library comprised of 20 new highly substituted spirooxindole derivatives.
T. Peňaška, K. Ormandyová, M. Mečiarová, J. Filo, R. Šebesta, New J. Chem. 2017, 41, 5506-5512; DOI: pubs.rsc.org/en/content/articlelanding/2017/nj/c7nj00189d#!divAbstract10.1039/C7NJ00189D.
T. Peňaška, K. Ormandyová, M. Mečiarová, J. Filo, R. Šebesta, New J. Chem. 2017, 41, 5506-5512; DOI: pubs.rsc.org/en/content/articlelanding/2017/nj/c7nj00189d#!divAbstract10.1039/C7NJ00189D.
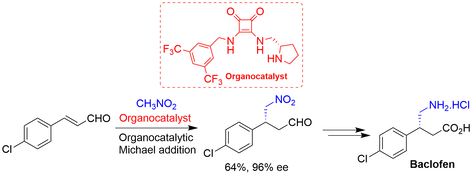
3-Substituted derivatives of γ-aminobutyric acid (GABA) are already certified, or potentially centrally-acting skeletal muscle relaxants and some of them are active ingredients in the drugs. Using the squaramide catalysis in a key step, baclofen was prepared in overall 25% yield and 96% ee.
E. Veverková, S. Bilka, R. Baran, R. Šebesta, Synthesis 2016, 48, 1474-1482; DOI: 10.1055/s-0035-1560420. (Featured on the cover of the issue!)
E. Veverková, S. Bilka, R. Baran, R. Šebesta, Synthesis 2016, 48, 1474-1482; DOI: 10.1055/s-0035-1560420. (Featured on the cover of the issue!)
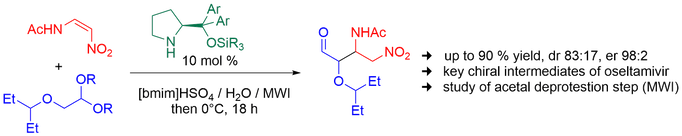
Synthesis of key chiral intermediates for oseltamivir and its derivatives from corresponding alkyloxyacetaldehyde acetals is described. Acetals are deprotected in the presence of acidic ionic liquid and water. These deprotection conditions are compatible with subsequent stereoselective Michael addition. A three-step sequence comprising acetal deprotection, Michael addition, and cyclization can be performed in a convenient one-pot operation.
P. Tisovský, T. Peňaška, M. Mečiarová, R. Šebesta, ACS Sust. Chem. Eng. 2015, 3, 3429-3434; DOI: 10.1021/acssuschemeng.5b01172.
P. Tisovský, T. Peňaška, M. Mečiarová, R. Šebesta, ACS Sust. Chem. Eng. 2015, 3, 3429-3434; DOI: 10.1021/acssuschemeng.5b01172.
Asymmetric dipolar cycloadditions as well as hetero-Diels-Alder reactions present an efficient method to synthesise heterocyclic products useful in pharmaceutical industry. Considerable disadvantage of organocatalytic dipolar cycloadditions is impractically long reaction times, which are necessary to obtain products in reasonable yields. Particularly, reactions of nitrones often last for several days. Use of ionic liquids, microwave and ultrasonic irradiation or other non-classical experimental techniques can have a positive impact on organocatalytic reactions.
M. Mojzesová, M. Mečiarová, A. Almássy, R. Marti, R. Šebesta, Chem. Pap. 2015, 69, 737-746. DOI: https://doi.org/10.1515/chempap-2015-0020
M. Mojzesová, M. Mečiarová, A. Almássy, R. Marti, R. Šebesta, Chem. Pap. 2015, 69, 737-746. DOI: https://doi.org/10.1515/chempap-2015-0020
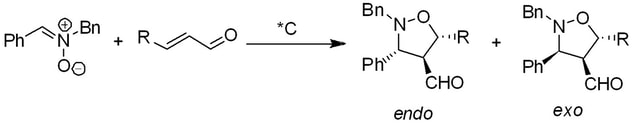
Chiral thioureas have found many applications as chiral catalysts. We have developed hydrogen bond-catalyzed aza-Mannich addition of azlactones (oxazol-5(4H)-ones) (1) to aldimines 2. The process benefits from the ability of bifunctional thioureas to affect the formation of α,β-diaminoacids precursors 3 in high yields and stereoselectivities. These compounds can be subsequently transformed into various α-substituted α,β-diaminoacids derivatives.
M. Žabka, A. Malastová, R. Šebesta RSC Adv. 2015, 5, 12890-12893. DOI: https://doi.org/10.1039/C5RA00092K
M. Žabka, A. Malastová, R. Šebesta RSC Adv. 2015, 5, 12890-12893. DOI: https://doi.org/10.1039/C5RA00092K
Michael addition of enolizable carbonyl compounds to nitroalkenes is a particularly appealing transformation, because it can serve in the synthesis of many bioactive molecules. One of them is pregabalin, a member of the gaba-pentinoid family of drugs used for the treatment of neuropathic pain and partial seizures. It is manufactured by a chemoenzymatic process. Organocatalytic approaches were also shown to be viable for pregabalin synthesis.
R. Baran, E. Veverková, A. Škvorcová, R. Šebesta, Org. Biomol. Chem. 2013, 11, 7705-7711.
DOI:https://doi.org/10.1039/C3OB41709C
R. Baran, E. Veverková, A. Škvorcová, R. Šebesta, Org. Biomol. Chem. 2013, 11, 7705-7711.
DOI:https://doi.org/10.1039/C3OB41709C
Oseltamivir phosphate is an active component of Tamiflu, one of the most potent antiviral drugs. It is currently produced via a 13 step synthesis. This synthesis suffer from potentially unreliable source of the starting material and involvement of hazardous procedures in the synthesis. Therefore, a great effort has been devoted to developing alternative syntheses. Use of asymmetric organocatalysis in the synthesis of oseltamivir has pioneered Hayashi and co-workers.
J. Rehák, M. Huťka, A. Latika, H. Brath, A. Almássy, V. Hajzer, J. Durmis, S. Toma, R. Šebesta, Synthesis 2012, 44, 2424-2430.
DOI: 10.1055/s-0031-1290396
J. Rehák, M. Huťka, A. Latika, H. Brath, A. Almássy, V. Hajzer, J. Durmis, S. Toma, R. Šebesta, Synthesis 2012, 44, 2424-2430.
DOI: 10.1055/s-0031-1290396