Fun with ferrocenes
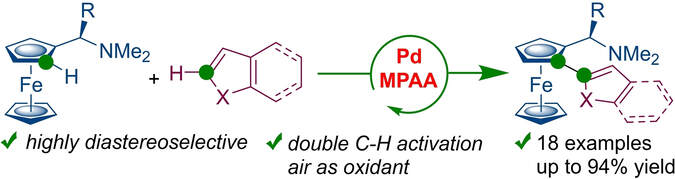
Pd-Complexes with N-Boc protected amino acids catalyze diastereoselective oxidative double C−H functionalization of chiral ferrocenyl amines with heteroarenes. A range of thiophene, pyrrole, furan and indole ferrocene derivative can be obtained in a single step and in high diastereomeric purities. C−H activation products can be transformed to corresponding phosphines. 19F NMR and DFT calculations helped elucidate reaction mechanism and reasons for stereoinduction.
K. Plevová, P. Kisszékelyi, D. Vargová, S. Andrejčák, V. Tóth, L. Fertáľ, E. Rakovský, J. Filo, R. Šebesta, Chem. Eur. J. 2021, 27, 15501-15507.
DOI: https://doi.org/10.1002/chem.202102624
K. Plevová, P. Kisszékelyi, D. Vargová, S. Andrejčák, V. Tóth, L. Fertáľ, E. Rakovský, J. Filo, R. Šebesta, Chem. Eur. J. 2021, 27, 15501-15507.
DOI: https://doi.org/10.1002/chem.202102624
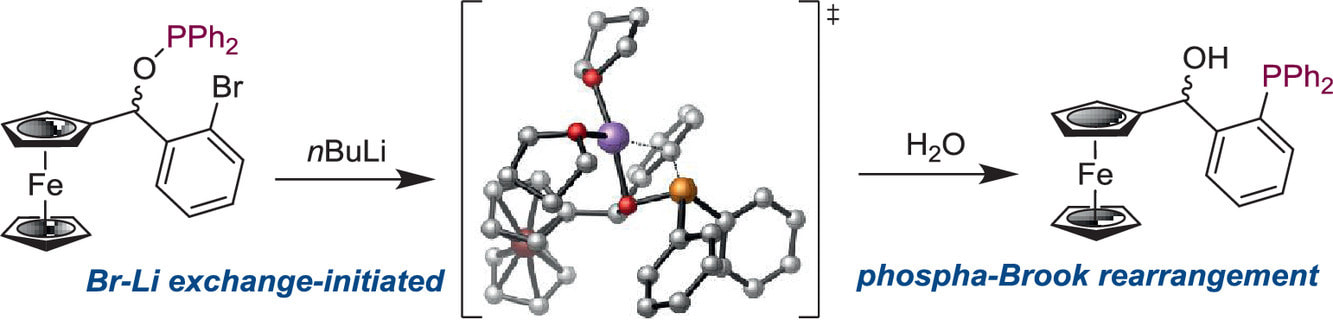
Ferrocene-containing phosphorus derivatives are important as ligands in transition-metal catalysis. We have investigated the synthesis of ferrocene-based phosphanes from corresponding phosphinites via retro-phospha-Brook rearrangement. Ferrocenyl phosphinites isomerize to thermodynamically more stable phosphine oxides. Interestingly, retro-phospha-Brook rearrangement proceeds on the adjacent phenyl ring with the assistance of the ferrocenyl moiety. DFT calculations provided insight into the reaction mechanisms of these transformations.
A. Almássy, A. Hejtmánková, D. Vargová, R. Šebesta, J. Organomet. Chem. 2021, 941, 121801.
DOI: https://doi.org/10.1016/j.jorganchem.2021.121801
A. Almássy, A. Hejtmánková, D. Vargová, R. Šebesta, J. Organomet. Chem. 2021, 941, 121801.
DOI: https://doi.org/10.1016/j.jorganchem.2021.121801
An efficient diastereoselective synthesis of planar chiral ferrocenes via Pd(II)-catalyzed direct C–H activation with arylboronic acids or pinacol esters is presented. The reaction was performed under mild conditions using commercially available achiral or chiral amino acids as ligands. The best results were obtained with (R)-Boc-alanine, which yielded products in 27–83% yield with diastereoselectivities ranging from 5:1 to 20:1 (11 examples). Diastereoisomeric products can also be obtained using (S)-Boc-alanine as a ligand. Stereoinduction of the reaction was explained by density functional theory calculations of possible transition states.
Diastereoselective Pd-Catalyzed CH Arylation of Ferrocenylmethanamines with Arylboronic Acids or Pinacol Esters. K. Plevová, B. Mudráková, E. Rakovský, R. Šebesta, J. Org. Chem. 2019, 84, 7312-7319; DOI: 10.1021/acs.joc.9b00953.
Diastereoselective Pd-Catalyzed CH Arylation of Ferrocenylmethanamines with Arylboronic Acids or Pinacol Esters. K. Plevová, B. Mudráková, E. Rakovský, R. Šebesta, J. Org. Chem. 2019, 84, 7312-7319; DOI: 10.1021/acs.joc.9b00953.
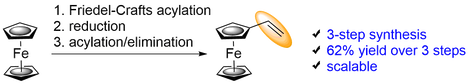
An improved, laboratory-friendly, short and efficient synthesis of vinylferrocene is reported. This three-step synthesis includes Friedel-Crafts acylation, reduction, and one-pot protection/deprotection acylation/elimination step to afford target compound in 62% yield over three steps.
K. Plevová, B. Mudráková, R. Šebesta, Synthesis, 2018, 50, 760-763; DOI: 10.1055/s-0036-1589142.
K. Plevová, B. Mudráková, R. Šebesta, Synthesis, 2018, 50, 760-763; DOI: 10.1055/s-0036-1589142.
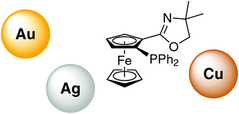
The coordination properties of phosphinoferrocene oxazolines with Group 11 metal ions were probed through a series of reactions between various metal precursors and the model ligand, rac-1-[4,5-dihydro-4,4-dimethyl-2-oxazolyl]-2-(diphenylphosphino)ferrocene (1). The reactions of 1 with CuX and AgCl produced the halide-bridged dimers [M(μ-X)(1-κ2N,P)], where M/X = Cu/Cl (2a), Cu/Br (2b), Cu/I (2c), and Ag/Cl (3), whereas the reaction with [AuCl(tht)] (tht = tetrahydrothiophene) yielded the chlorogold(I) complex [AuCl(1-κP)] (4). When metal precursors without strongly coordinating halide ligands were used (viz., [Cu(MeCN)4][PF6] and AgClO4), the complexation reactions generated bis-chelate complexes [M(1-κ2N,P)2]X (5: M/X = Cu/PF6, 6: M/X = Ag/ClO4). A similar reaction with [Au(tht)2]ClO4 produced [Au(1-κP)2]ClO4 (7), wherein the gold centre is linearly coordinated by two phosphine moieties. All complexes except for 4 were structurally authenticated by X-ray crystallography. The observed coordination behaviour and structures of the isolated complexes are discussed in the context of the catalytic properties of chiral ligands structurally related to the model compound 1.
O. Bárta, M. Drusan, I. Císařová, R. Šebesta, P. Štěpnička, New J. Chem. 2018, 42, 11450-11457; DOI: 10.1039/C8NJ02101E.
O. Bárta, M. Drusan, I. Císařová, R. Šebesta, P. Štěpnička, New J. Chem. 2018, 42, 11450-11457; DOI: 10.1039/C8NJ02101E.
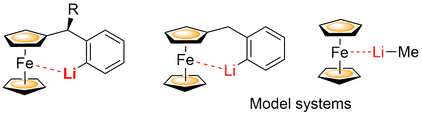
Intermetallic Fe-Li interaction likely occurs during some stereoselective lithiation reactions of ferrocene derivatives. The nature of this interaction was assessed on a model system of ferrocene – methyl lithium complex using coupled cluster methods CCSD(T). CCSD(T) calculations predicted interaction energy of -38.0 kJ.mol–1 and Fe-Li distance of 2.75 Å. The ability of commonly used DFT functionals to describe this interaction was also screened. The best agreement of structural parameters (energy, and Fe-Li distance) with the CCSD(T) calculated results was achieved by dispersion corrected TPSS functional. DFT calculations were also used for natural bond orbital analysis of the intramolecular Fe-Li interaction in benzylferrocene ortho-lithiated on the phenyl ring. This bimetallic bond can be explained as a donor-acceptor orbital interactions between the iron lone pair or C-Fe bonding electrons with the empty orbitals of lithium.
A. Malastová, J. Vallo, A. Almássy, M. Pitoňák, P. Neogrády, R. Šebesta, Eur. J. Inorg. Chem. 2017, 483-488; DOI: 10.1002/ejic.201601114.
A. Malastová, J. Vallo, A. Almássy, M. Pitoňák, P. Neogrády, R. Šebesta, Eur. J. Inorg. Chem. 2017, 483-488; DOI: 10.1002/ejic.201601114.